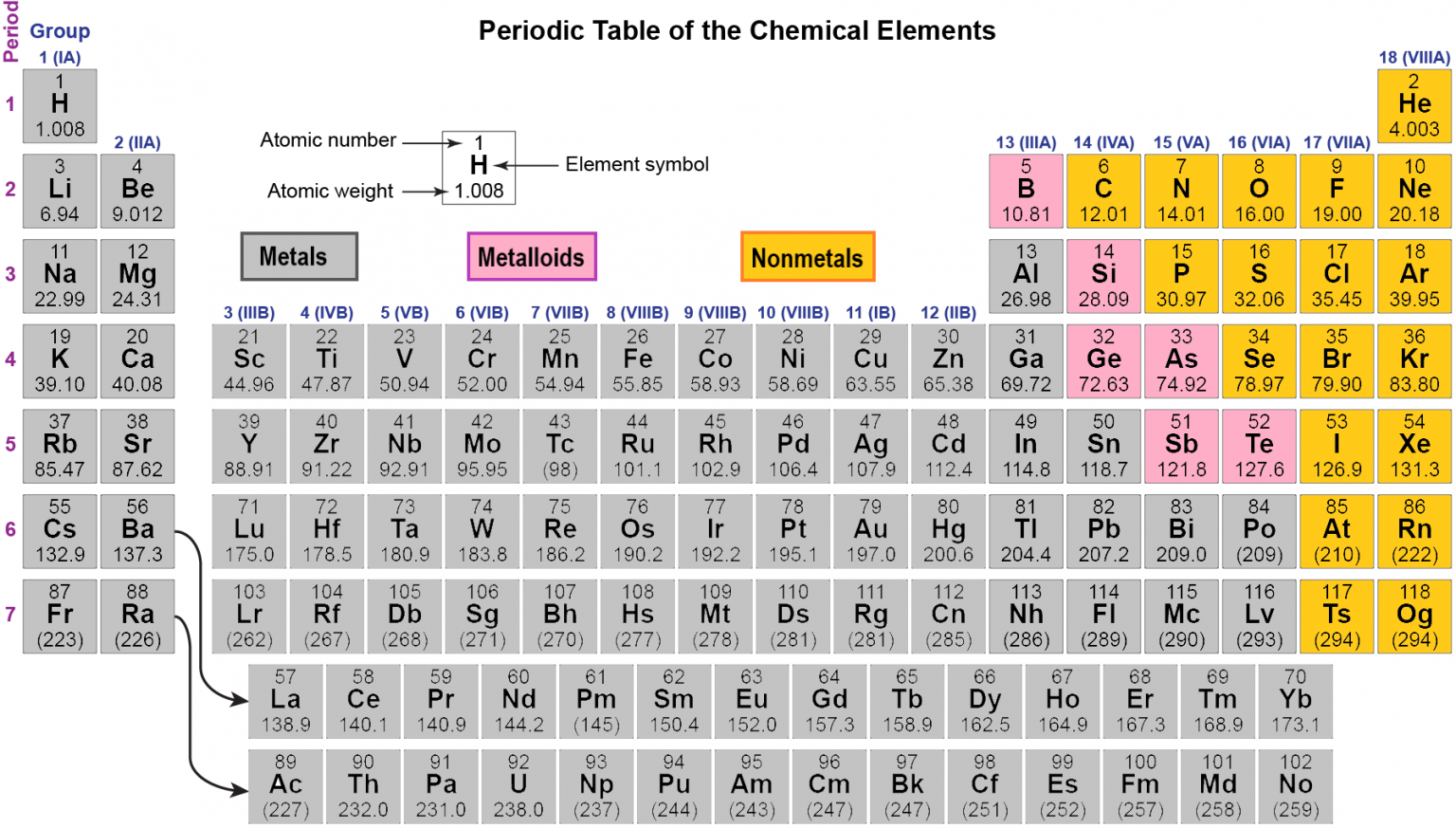
As expected, semimetals exhibit properties intermediate between metals and nonmetals. Notice the heavier line running diagonally from boron. For example, an electronegativity periodic table color codes the elements based on how electronegative they are. Periodic tables can use color to identify other element characteristics. Color coding helps identify similar elements such as these at a glance. Metalloids are shaded lavender and nonmetals are rose. The metalloids and nonmetals, for example, don't fall neatly into the same column. Artificial elements are denoted by the outlined symbols. elements f-block FIGURE 7.1 Lanthanides Actinides The main ways in which we divide the periodic table into different sections. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. The unabridged version of the Periodic Table of the Elements, showing all 118 elements and emphasizing the fact that the vast majority of them are metals (shaded blue). Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. 
Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. Nonmetals often have a drab look, low melting temperatures, boiling boils, and densities, and are poor heat and. Metals are excellent heat and electricity conductors, as well as malleable (able to be hammered into sheets) and ductile materials (can be drawn into wire). The vast majority of the known elements are metals. Answer: Elements are made up of metals, non-metals, and metalloids. 
Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Metalloids are elements that look like metals and in some ways behave like metals but also have some nonmetallic properties. The key for this activity is located as the preview. Just as shown in the table above, metals and nonmetals on the periodic table are often separated by a stairstep diagonal line, and several elements near this line are often called metalloids (Si, Ge, As, Sb, Te, and At). The 18 columns are labeled Group and the 7 rows are labeled Period. Metals are excellent heat and electrical conductors. A metal is a hard solid substance with a lustrous sheen that conducts heat and electricity. The periodic table may be further color-coded to include other groups such as transition metals, rare earths, alkali. The elements are placed in the periodic table and classified as metals, non-metals, or metalloids, based on their groupings. In general, the color blue is used for metals, orange for metalloids and green for non-metals. This color represents the classes of elements, which include metals, metalloids and non-metals. Students will need three colored pencils to complete this activity. In a color-coded periodic table of the elements, the box for each element is colored. Such a treatment has been obstructed by the traditional view of metalloids as in-between elements understandable but needless boundary squabbles and a group-by-group view of the reactive nonmetals. Learn the major parts of the periodic table by coloring the metals, nonmetals, and metalloids. The periodic table can be simply demarcated into four classes of metal and four classes of nonmetal. Ideas for Using Activity: The key can be color printed and laminated to make a class set. All the elements in the periodic table are classified into three categories namely Metals, Non-Metals, and Metalloids. Repeat the process again for coding solids, liquids, and gases at room temperature. Metalloids are the elements in the Chemistry Periodic Table whose properties lie in between those of metals and non-metals. Remind students to fill in the key with the colors they chose for each. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. Periodic Table : Color Metals, Nonmetals, & Metalloids (Pt-4) Created by. Directions: Instruct students to color code metals, nonmetals, and metalloids on the periodic table. \): Antimony.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
